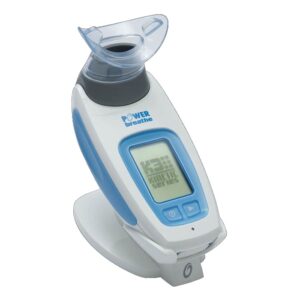
The KHP2 inspiratory muscle training device (IMT) for respiratory muscle training (RMT) programs has been developed to be used by patients where lung function is limited. It can be easily used in the home as part of on-going rehabilitation or even prior to surgery for pre-operative conditioning.
Findings from clinical research reveal good adherence to training because the patient can see their progress on-screen. Thirty-six training sessions can be stored and reviews, and it’s because of this the research showed a high compliance of 90% .
The POWERbreathe KHP2 is drug-free, suitable for almost anyone and should cause no harmful side effects when used properly.
How does it work?
The POWERbreathe KH series uses piezo-resistive pressure sensing technology for precise measurement of respiratory pressure 500 times/second. The triple-aperture rotary valve system makes up to 4000 precision movements every second.
The POWERbreathe KHP2 uses an electronically controlled, rapid-response valve to create a resistance to inhalation. Training against this resistance causes the inspiratory muscles to adapt, becoming stronger and more resistant to fatigue. This leads to reduced breathlessness and improved exercise tolerance and quality of life. As the patient breathes through the POWERbreathe KH2 you will notice that they gradually have to work harder to breathe in. This is the effect of resistance training acting on the inspiratory muscles (primarily the diaphragm and intercostal muscles). When breathing out, there is no resistance and the patient should breathe out normally, allowing the chest and breathing muscles to relax, naturally pushing the air from their lungs.
The POWERbreathe KHP2 training resistance is specifically designed to match the dynamic changes in breathing muscle strength throughout the breath and can automatically adapt to increases in inspiratory muscle strength at the beginning of each training session. Results are displayed on screen following a training or assessment session, or can be viewed in real-time using the Breathe-Link Medic PC software supplied. Parameters displayed include Maximal Inspiratory Pressure (cmH2O, highest drug-free average), Peak Inspiratory Flow (L/s), Training load (cmH2O), Average Power (Watts), Average inhaled volume (L) and Energy (Joules).
Training load is gradually introduced over the first five breaths of a training session. The first two breaths are unloaded. During these breaths, inhaled volume and flow are measured and used to set an appropriate training load. Loading is then gradually introduced during breaths three and four until full loading is achieved for breath five and onwards. The POWERbreathe KHP2 training regime of 30 breaths, twice a day typically takes only a few minutes a day and, used properly, patients should start to observe the benefits within just a few weeks.
Precautions & Contraindications
POWERbreathe IMT is drug free, suitable for almost anyone and should cause no harmful side effects when used properly. Please read the following precautions and contraindications to ensure that POWERbreathe IMT is used safely and appropriately.
Precautions:
- The patient must not make changes to any prescribed medication or prescribed treatment programme without consulting their doctor.
- A patient must not use the POWERbreathe IMT device whilst taking part in other activities where they may become distracted, such as walking, running and driving.
- POWERbreathe IMT should not be used if the patient is suffering from a ruptured eardrum or any other condition of the eardrum.
- It is not recommended to use POWERbreathe IMT if the patient has suffered from or are likely to suffer from Costochondritis.
- To prevent the potential transmission of infections, we recommend that POWERbreathe IMT is not shared with other users, including family members.
- This product is not intended to diagnose, treat, cure or prevent any disease.
- POWERbreathe IMT is designed for exercising the inspiratory muscles only. No other use is intended or implied.
- Anyone who is under the age of 16 should only use POWERbreathe IMT with supervision from an adult. POWERbreathe IMT contains small parts and is not suitable for children under 7 years.
- Whilst training with POWERbreathe IMT, the patient should feel resistance when inhaling but it should not be painful. If the patient feels pain whilst using POWERbreathe IMT, they should stop immediately and consult their doctor.
- Some users may experience slight ear discomfort when training with POWERbreathe IMT, especially if they are recovering from a cold. This is caused by inadequate equalisation of pressure between the mouth and ears. If symptoms persist, patients should consult their doctor.
- If a patient is suffering from a cold, sinusitis or respiratory tract infection, we advise that they do not use POWERbreathe IMT until symptoms have disappeared.
- If a patient starts to feel dizzy using POWERbreathe IMT, please recommend that they pause until recovered and take longer gaps between breaths.
The Healthcare Professional should take into consideration the following conditions before patients use POWERbreathe IMT:
- A history of spontaneous pneumothorax (a collapsed lung that was not due to traumatic injury e.g. broken rib), as it may lead to a recurrence of the condition.
- Pulmonary hypertension or large bullae on chest x-ray.
- Marked osteoporosis with history of rib fractures.
- Desaturation during or following IMT (<94%).
Contraindications
Inspiratory muscle training, such as training with POWERbreathe, is not recommended for patients with certain conditions, including the following:
- Patients who have undergone recent abdominal surgery and those with an abdominal hernia.
- Asthma patients who have a very low symptom perception and suffer from frequent, severe exacerbations or with an abnormally low perception of dyspnoea
- If a patient is suffering from a ruptured eardrum or any other condition of the ear
- Patients with marked elevated left ventricular end-diastolic volume and pressure
- Patients with worsening heart failure signs and symptoms after RMT / IMT
- If a patient is suffering from a cold, sinusitis or respiratory tract infection, we advise that they do not use their POWERbreathe IMT device.
Contraindications
Inspiratory muscle training, such as training with POWERbreathe, creates a negative pressure inside the chest, throat, ears and sinuses and is not recommended for patients with certain conditions, including the following:
- Asthma patients who have low symptom perception and suffer from frequent, severe exacerbations or with an abnormally low perception of dyspnoea
- If a patient is suffering from a ruptured eardrum or any other condition of the ear
- Patients with marked elevated left ventricular end-diastolic volume and pressure
- Patients with worsening heart failure signs and symptoms after RMT / IMT
- A collapsed lung due to a traumatic injury that has not healed fully







Reviews
There are no reviews yet.